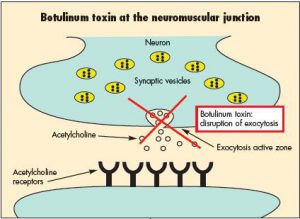
Heptavalent Botulinum Antitoxin Treatment
August 3rd, 2019Jason S Richardson, Geraldine S Parrera, Hugo Astacio, Harpreet Sahota, Deborah M Anderson, Christine Hall, Tim Babinchak, Safety and Clinical Outcomes of an Equine-Derived Heptavalent Botulinum Antitoxin Treatment for Confirmed or Suspected Botulism in the United States, Clinical Infectious Diseases, , ciz515, https://doi.org/10.1093/cid/ciz515
“…….Among 162 patients, 7 (4.3%) experienced BAT product-related serious adverse events, including 1 (0.6%) report each of pneumonia, pneumonia aspiration, ventricular tachycardia, upper gastrointestinal hemorrhage, anaphylactic reaction, acute kidney injury, and acute myocardial infarction. Thirty-one (19.1%) patients had 41 BAT product-related adverse events. Six (3.7%) deaths were reported in the registry. All deaths were attributed to the underlying illness and were assessed as unlikely related to BAT product. Among 113 (69.8%) patients with a final diagnosis of botulism, those treated early (≤2 days) spent fewer days in the hospital (5 vs 15.5 days), in the intensive care unit (4 vs 12 days), and on mechanical ventilation (6 vs 14.5 days) than those treated late (>2 days), respectively……..”