Archive for the ‘Malaria’ Category
Experimental PfSPZ malaria vaccine provides durable protection against multiple strains in NIH clinical trial
Thursday, February 23rd, 2017Tuesday, February 21, 2017
Experimental PfSPZ malaria vaccine provides durable protection against multiple strains in NIH clinical trial
“An effective malaria vaccine will need to protect people living in endemic areas against multiple strains of the mosquito-borne disease.”
—Anthony S. Fauci, M.D., Director, NIAID
An investigational malaria vaccine has protected a small number of healthy U.S. adults from infection with a malaria strain different from that contained in the vaccine, according to a study published today in the Proceedings of the National Academy of Sciences (PNAS). The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, sponsored and co-conducted the Phase 1 clinical trial.
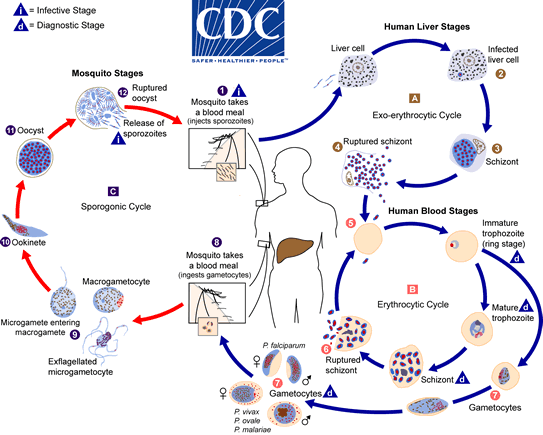
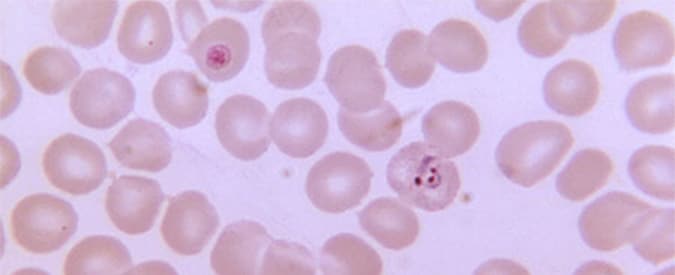
Malaria is transmitted to humans through the bite of infected mosquitoes, which inject immature malaria parasites called sporozoites into a person’s bloodstream. The parasites travel to the liver, where they mature, multiply and spread via the bloodstream throughout the body causing malaria symptoms including chills, fever, headache, nausea, sweating and fatigue. According to the World Health Organization, 214 million people were infected with malaria globally in 2015 and 438,000 people died, mostly young African children. The species Plasmodium falciparum is the most common cause of malaria morbidity and mortality in Africa. In the United States, travel-related malaria is a concern for international tourists, aid workers and military personnel worldwide.
The PfSPZ Vaccine used in this study was developed by Sanaria Inc., of Rockville, Maryland. The vaccine contains weakened P. falciparum sporozoites that do not cause infection but are able to generate a protective immune response against live malaria infection. Earlier research at the NIH Clinical Center with the PfSPZ Vaccine found it to be safe, well-tolerated and protective for more than a year when tested in healthy U.S. adults against a single Africa-derived malaria strain matched to the PfSPZ Vaccine.
“An effective malaria vaccine will need to protect people living in endemic areas against multiple strains of the mosquito-borne disease,” said NIAID Director Anthony S. Fauci, M.D. “These new findings showing cross-protection with the PfSPZ Vaccine suggest that it may be able to accomplish this goal.”
The study enrolled 31 healthy adults ages 18 to 45 years, and was led by Julie E. Ledgerwood, D.O. of NIAID’s Vaccine Research Center (VRC), and Kirsten E. Lyke, M.D. of the University of Maryland Center for Vaccine Development in Baltimore. Participants were assigned to receive three doses of the PfSPZ Vaccine at eight-week intervals by rapid intravenous injection.
Nineteen weeks after receiving the final dose of the test vaccine, participants who received the vaccine and a group of non-vaccinated volunteers were exposed in a controlled setting to bites from mosquitoes infected with the same strain of P. falciparum parasites (NF54, from Africa) that were used to manufacture PfSPZ Vaccine.
Nine of the 14 participants (64 percent) who received PfSPZ Vaccine demonstrated no evidence of malaria parasites; all six of the non-vaccinated participants who were challenged at the same time had malaria parasites in their blood.
Of the nine participants who showed no evidence of malaria, six participants were again exposed in a controlled setting to mosquito bites, this time from mosquitoes infected with a different strain of P. falciparum parasite, 33 weeks after the final immunization. In this group, 5 of the 6 participants (83 percent) were protected against malaria infection; none of the six participants who did not receive the vaccine and were challenged were protected. All participants who became infected with malaria immediately received medical treatment.
“Achieving durable protection against a malaria strain different from the vaccine strain, over eight months after vaccination, is an indication of this vaccine’s potential,” said Robert A. Seder, M.D., chief of the Cellular Immunology Section of NIAID’s Vaccine Research Center and senior author of the PNAS paper. “If we can build on these findings with the PfSPZ Vaccine and induce higher efficacy, we may be on our way to a vaccine that could effectively protect people against a variety of malaria parasites where the disease is prevalent.”
The research team found that the PfSPZ Vaccine activated T cells, a key component of the body’s defenses against malaria, and induced antibody responses in all vaccine recipients. Vaccine-specific T-cell responses were comparable when measured against both of the malaria challenge strains, providing some insights into how the vaccine was mediating protection.
Ongoing research will determine whether protective efficacy can be improved by changes to the PfSPZ Vaccine dose and number of immunizations. Accordingly, a Phase 2 efficacy trial testing three different dosages in a three-dose vaccine regimen is now underway in 5-to 12-month-old infants in Western Kenya to assess safety and efficacy against natural infection.
Sanaria Inc., designed, manufactured, and provided PfSPZ Vaccine and the heterologous challenge mosquitoes. NIAID supported the development of the experimental vaccine through several Small Business Innovation Research grants: 5R44AI055229-11, 5R44AI058499-08, and 5R44AI058375-08. For more information about the Phase 1 study, see clinicaltrials.gov using the identifier: NCT02015091.
NIAID conducts and supports research — at NIH, throughout the United States, and worldwide — to study the causes of infectious and immune-mediated diseases, and to develop better means of preventing, diagnosing and treating these illnesses. News releases, fact sheets and other NIAID-related materials are available on the NIAID website.
About the National Institutes of Health (NIH): NIH, the nation’s medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit www.nih.gov.
NIH…Turning Discovery Into Health®
Reference
K. Lyke et al. PfSPZ vaccine induces strain-transcending T cells and durable protection against heterologous controlled human malaria infection. PNAS DOI 10.1073/pnas.1615324114 (2017).
A malaria vaccine was completely successful in preventing the disease in 35 humans
Thursday, February 16th, 2017Sterile protection against human malaria by chemoattenuated PfSPZ vaccine
Nature Year published:(2017)
DOI:doi:10.1038/nature21060
“…..PfSPZ-CVac is a highly efficacious vaccine candidate; when we are able to optimize the immunization regimen (dose, interval between doses, and drug partner), this vaccine could be used for combination mass drug administration and a mass vaccination program approach to eliminate malaria from geographically defined areas……”
Artemether-lumefantrine: A key malaria treatment has failed for the first time in 4 patients being treated in the UK
Tuesday, January 31st, 2017Malaria: Peru reports an increase in the incidence of the disease with more than 60,000 cases in 2015.
Thursday, January 12th, 2017“…Malaria is a parasitic disease, caused by a Plasmodium parasite, transmitted by the bite of a mosquito already infected with malaria. Only a few species of anophelines are able to transmit the parasite. In the Amazon basin, Anopheles darlingi is the major vector of the disease. …”
Cerebral Malaria: New study reveals the processes that lead to fatal outcomes
Wednesday, December 14th, 2016“…..as red blood cells containing the parasite adhere to cerebral blood vessels (a hallmark of Cerebral Malaria), the immune system attempts to clean them off. Despite these efforts, endothelial cells making up the walls of cerebral blood vessels shed bits of the parasite, which CD8+ T cells recognize, causing those immune cells to attach to and attack the vessels. Once the CD8+T cells amassed on the surface of brain blood vessels, the vessels began to leak. The subsequent leaking led to swelling and increased pressure in the brain, which was fatal. ….”
WHO’s World Malaria Report 2016
Wednesday, December 14th, 201613 December 2016 | GENEVA – WHO’s World Malaria Report 2016 reveals that children and pregnant women in sub-Saharan Africa have greater access to effective malaria control. Across the region, a steep increase in diagnostic testing for children and preventive treatment for pregnant women has been reported over the last 5 years. Among all populations at risk of malaria, the use of insecticide-treated nets has expanded rapidly.
But in many countries in the region, substantial gaps in programme coverage remain. Funding shortfalls and fragile health systems are undermining overall progress, jeopardizing the attainment of global targets.
Scale-up in malaria control
Sub-Saharan Africa carries a disproportionately high share of the global malaria burden. In 2015, the region was home to 90% of malaria cases and 92% of malaria deaths. Children under five years of age are particularly vulnerable, accounting for an estimated 70% of all malaria deaths.
Diagnostic testing enables health providers to rapidly detect malaria and prescribe life-saving treatment. New findings presented in the report show that, in 2015, approximately half (51%) of children with a fever seeking care at a public health facility in 22 African countries received a diagnostic test for malaria, compared to 29% in 2010.
To protect women in areas of moderate and high malaria transmission in Africa, WHO recommends “intermittent preventive treatment in pregnancy” (IPTp) with sulfadoxine-pyrimethamine. The treatment, administered at each scheduled antenatal care visit after the first trimester, can prevent maternal and infant mortality, anaemia, and the other adverse effects of malaria in pregnancy.
According to available data, there was a five-fold increase in the percentage of women receiving the recommended 3 or more doses of this preventive treatment in 20 African countries. Coverage reached 31% in 2015, up from 6% in 2010.
Insecticide-treated nets are the cornerstone of malaria prevention efforts in Africa. The report found that more than half (53%) of the population at risk in sub-Saharan Africa slept under a treated net in 2015, compared to 30% in 2010.
Last month, WHO released the findings of a major 5-year evaluation in 5 countries. The study showed that people who slept under long-lasting insecticidal nets (LLINs) had significantly lower rates of malaria infection than those who did not use a net, even though mosquitoes showed resistance to pyrethroids (the only insecticide class used in LLINs) in all of these areas.
An unfinished agenda
Malaria remains an acute public health problem, particularly in sub-Saharan Africa. According to the report, there were 212 million new cases of malaria and 429 000 deaths worldwide in 2015.
There are still substantial gaps in the coverage of core malaria control tools. In 2015, an estimated 43% of the population in sub-Saharan Africa was not protected by treated nets or indoor spraying with insecticides, the primary methods of malaria vector control.
In many countries, health systems are under-resourced and poorly accessible to those most at risk of malaria. In 2015, a large proportion (36%) of children with a fever were not taken to a health facility for care in 23 African countries.
“We are definitely seeing progress,” notes Dr Pedro Alonso, Director of the WHO Global Malaria Programme. “But the world is still struggling to achieve the high levels of programme coverage that are needed to beat this disease.”
Global targets
At the 2015 World Health Assembly, Member States adopted the Global Technical Strategy for Malaria 2016-2030. The Strategy set ambitious targets for 2030 with milestones every 5 years to track progress.
Eliminating malaria in at least 10 countries is a milestone for 2020. The report shows that prospects for reaching this target are bright: In 2015, 10 countries and territories reported fewer than 150 indigenous cases of malaria, and a further 9 countries reported between 150 and 1000 cases.
Countries that have achieved at least 3 consecutive years of zero indigenous cases of malaria are eligible to apply for the WHO certification of malaria elimination. In recent months, the WHO Director-General certified that Kyrgyzstan and Sri Lanka had eliminated malaria.
But progress towards other key targets must be accelerated. The Strategy calls for a 40% reduction in malaria case incidence by the year 2020, compared to a 2015 baseline. According to the report, less than half (40) of the 91 countries and territories with malaria are on track to achieve this milestone. Progress has been particularly slow in countries with a high malaria burden.
An urgent need for more funding
Sustained and sufficient funding for malaria control is a serious challenge. Despite a steep increase in global investment for malaria between 2000 and 2010, funding has since flat-lined. In 2015, malaria funding totalled US$ 2.9 billion, representing only 45% of the funding milestone for 2020 (US$ 6.4 billion).
Governments of malaria-endemic countries provided about 31% of total malaria funding in 2015. The United States of America is the largest international malaria funder, accounting for about 35% of total funding in 2015, followed by the United Kingdom of Great Britain and Northern Ireland (16%).
If global targets are to be met, funding from both domestic and international sources must increase substantially.
Note to editors
RTS,S/AS01 malaria vaccine
Last month, WHO announced that the world’s first malaria vaccine would be rolled out through pilot projects in 3 countries in sub-Saharan Africa. Vaccinations will begin 2018. The vaccine, known as RTS,S, acts against P. falciparum, the most deadly malaria parasite globally, and the most prevalent in Africa. Advanced clinical trials have shown RTS,S to provide partial protection against malaria in young children.
WHO multi-country evaluation on LLINs
On 16 November 2016, WHO released the findings of a 5-year evaluation conducted in 340 locations across 5 countries: Benin, Cameroon, India, Kenya and Sudan. The findings of this study reaffirm the WHO recommendation of universal LLIN coverage for all populations at risk of malaria.
For more information, please contact:
Saira Stewart
Technical officer, WHO
Mobile: +41 79 500 6538
Email: stewarts@who.int
Gregory HÄRTL
Coordinator, News, Social Media and Monitoring
Telephone: +41 22 791 4458
Mobile: +41 79 203 6715
Email: hartlg@who.int
Efficacy of topical mosquito repellent (picaridin) and long-lasting insecticidal nets for control of malaria.
Friday, July 1st, 2016Efficacy of RTS, S/AS01, the first vaccine developed to fight malaria.
Friday, July 1st, 2016“…A three-dose vaccination with RTS,S/AS01 was initially protective against clinical malaria, but this result was offset by rebound in later years in areas with higher-than-average exposure to malaria parasites….”