WHO
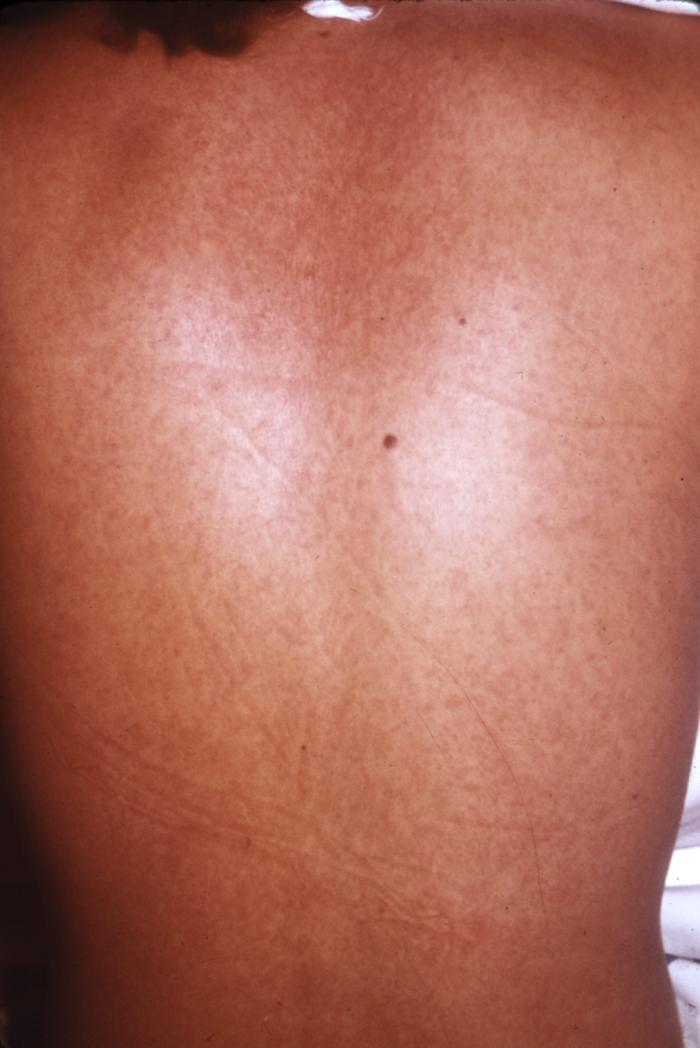
CDC/ Conrad: This type of maculopapular rash, which can appear on Marburg patients around the fifth day after the onset of symptoms, usually may be found on the patient’s chest, back and stomach. This patient’s skin blanched under pressure, which is a common characteristic of a Marburg virus rash.
Updated October 2017
Key facts
- Marburg virus disease (MVD), formerly known as Marburg haemorrhagic fever, is a severe, often fatal illness in humans.
- Rousettus aegyptiacus, fruit bats of the Pteropodidae family, are considered to be natural hosts of Marburg virus. The Marburg virus is transmitted to people from fruit bats and spreads among humans through human-to-human transmission.
- The Marburg virus causes severe viral haemorrhagic fever in humans.
- The average MVD case fatality rate is around 50%. Case fatality rates have varied from 24% to 88% in past outbreaks depending on virus strain and case management.
- Community engagement is key to successfully controlling outbreaks. Good outbreak control relies on applying a package of interventions, namely case management, infection prevention and control practices, surveillance and contact tracing, a good laboratory service, safe burials and social mobilization.
- Early supportive care with rehydration, symptomatic treatment improves survival. There is as yet no licensed treatment proven to neutralize the virus but a range of blood products, immune therapies and drug therapies are currently under development.
Marburg virus is the causative agent of Marburg virus disease (MVD), a disease with a case fatality ratio of up to 88%. Marburg virus disease was initially detected in 1967 after simultaneous outbreaks in Marburg and Frankfurt in Germany; and in Belgrade, Serbia.
Marburg and Ebola viruses are both members of the Filoviridae family (filovirus). Though caused by different viruses, the two diseases are clinically similar. Both diseases are rare and have the capacity to cause dramatic outbreaks with high fatality rates.
Two large outbreaks that occurred simultaneously in Marburg and Frankfurt in Germany, and in Belgrade, Serbia, in 1967, led to the initial recognition of the disease. The outbreak was associated with laboratory work using African green monkeys (Cercopithecus aethiops) imported from Uganda. Subsequently, outbreaks and sporadic cases have been reported in Angola, Democratic Republic of the Congo, Kenya, South Africa (in a person with recent travel history to Zimbabwe) and Uganda. In 2008, two independent cases were reported in travelers who had visited a cave inhabited by Rousettus bat colonies in Uganda.
Transmission
Initially, human MVD infection results from prolonged exposure to mines or caves inhabited by Rousettus bat colonies.
Marburg spreads through human-to-human transmission via direct contact (through broken skin or mucous membranes) with the blood, secretions, organs or other bodily fluids of infected people, and with surfaces and materials (e.g. bedding, clothing) contaminated with these fluids.
Health-care workers have frequently been infected while treating patients with suspected or confirmed MVD. This has occurred through close contact with patients when infection control precautions are not strictly practiced. Transmission via contaminated injection equipment or through needle-stick injuries is associated with more severe disease, rapid deterioration, and, possibly, a higher fatality rate.
Burial ceremonies that involve direct contact with the body of the deceased can also contribute in the transmission of Marburg.
People remain infectious as long as their blood contains the virus.
Sexual transmission
Marburg virus transmission via infected semen has been documented up to seven weeks after clinical recovery. More surveillance data and research are needed on the risks of sexual transmission, and particularly on the prevalence of viable and transmissible virus in semen over time. In the interim, and based on present evidence, WHO recommends that:
- All Marburg survivors and their sexual partners should receive counselling to ensure safer sexual practices until their semen has twice tested negative for Marburg virus.
- Survivors should be provided with condoms.
- Male Marburg survivors should be enrolled in semen testing programmes when discharged (starting with counselling) and offered semen testing when mentally and physically ready, within three months of disease onset.
- Marburg survivors and their sexual partners should either:
- abstain from all sexual practices, or
- observe safer sexual practices through correct and consistent condom use until their semen has twice tested undetected ( negative) for Marburg virus.
- Having tested undetected (negative), survivors can safely resume normal sexual practices with minimized risk of Marburg virus transmission.
- Male survivors of Marburg virus disease should practice safer sexual practices and hygiene for 12 months from onset of symptoms or until their semen twice tests undetected (negative) for Marburg virus.
- Until such time as their semen has twice tested undetected (negative) for Marburg, survivors should practice good hand and personal hygiene by immediately and thoroughly washing with soap and water after any physical contact with semen, including after masturbation. During this period used condoms should be handled safely, and safely disposed of, so as to prevent contact with seminal fluids.
- All survivors, their partners and families should be shown respect, dignity and compassion.
Symptoms of Marburg virus disease
The incubation period (interval from infection to onset of symptoms) varies from 2 to 21 days.
Illness caused by Marburg virus begins abruptly, with high fever, severe headache and severe malaise. Muscle aches and pains are a common feature. Severe watery diarrhoea, abdominal pain and cramping, nausea and vomiting can begin on the third day. Diarrhoea can persist for a week. The appearance of patients at this phase has been described as showing “ghost-like” drawn features, deep-set eyes, expressionless faces, and extreme lethargy. In the 1967 European outbreak, non-itchy rash was a feature noted in most patients between 2 and 7 days after onset of symptoms.
Many patients develop severe haemorrhagic manifestations between 5 and 7 days, and fatal cases usually have some form of bleeding, often from multiple areas. Fresh blood in vomitus and faeces is often accompanied by bleeding from the nose, gums, and vagina. Spontaneous bleeding at venepuncture sites (where intravenous access is obtained to give fluids or obtain blood samples) can be particularly troublesome. During the severe phase of illness, patients have sustained high fevers. Involvement of the central nervous system can result in confusion, irritability, and aggression. Orchitis (inflammation of one or both testicles) has been reported occasionally in the late phase of disease (15 days).
In fatal cases, death occurs most often between 8 and 9 days after symptom onset, usually preceded by severe blood loss and shock.
Persistent virus in people recovering from Marburg virus disease
Marburg virus is known to persist in immune-privileged sites in some people who have recovered from Marburg virus disease. These sites include the testicles and the inside of the eye.
- In women who have been infected while pregnant, the virus persists in the placenta, amniotic fluid and fetus.
- In women who have been infected while breastfeeding, the virus may persist in breast milk.
Relapse-symptomatic illness in the absence of re-infection in someone who has recovered from MVD is a rare event, but has been documented. Reasons for this phenomenon are not yet fully understood.
Diagnosis
It can be difficult to clinically distinguish MVD from other infectious diseases such as malaria, typhoid fever, shigellosis, meningitis and other viral haemorrhagic fevers. Confirmation that symptoms are caused by Marburg virus infection are made using the following diagnostic methods:
- antibody-capture enzyme-linked immunosorbent assay (ELISA)
- antigen-capture detection tests
- serum neutralization test
- reverse transcriptase polymerase chain reaction (RT-PCR) assay
- electron microscopy
- virus isolation by cell culture.
Samples collected from patients are an extreme biohazard risk; laboratory testing on non-inactivated samples should be conducted under maximum biological containment conditions. All biological specimens should be packaged using the triple packaging system when transported nationally and internationally.
Treatment and vaccines
Supportive care – rehydration with oral or intravenous fluids – and treatment of specific symptoms, improves survival. There is as yet no proven treatment available for MVD. However, a range of potential treatments including blood products, immune therapies and drug therapies are currently being evaluated.
Marburg virus in animals
Rousettus aegyptiacus bats are considered natural hosts for Marburg virus. There is no apparent disease in the fruit bats. As a result, the geographic distribution of Marburg virus may overlap with the range of Rousettus bats.
African green monkeys (Cercopithecus aethiops) imported from Uganda were the source of infection for humans during the first Marburg outbreak.
Experimental inoculations in pigs with different Ebola viruses have been reported and show that pigs are susceptible to filovirus infection and shed the virus. Therefore pigs should be considered as a potential amplifier host during MHF outbreaks. Although no other domestic animals have yet been confirmed as having an association with filovirus outbreaks, as a precautionary measure they should be considered as potential amplifier hosts until proven otherwise.
Precautionary measures are needed in pig farms in Africa to avoid pigs becoming infected through contact with fruit bats. Such infection could potentially amplify the virus and cause or contribute to MHF outbreaks.
Prevention and control
Good outbreak control relies on applying a package of interventions, namely case management, surveillance and contact tracing, a good laboratory service, safe and dignified burials, and social mobilization. Community engagement is key to successfully controlling outbreaks. Raising awareness of risk factors for Marburg infection and protective measures that individuals can take is an effective way to reduce human transmission.
Risk reduction messaging should focus on several factors:
- Reducing the risk of bat-to-human transmission arising from prolonged exposure to mines or caves inhabited by fruit bat colonies. During work or research activities or tourist visits in mines or caves inhabited by fruit bat colonies, people should wear gloves and other appropriate protective clothing (including masks). During outbreaks all animal products (blood and meat) should be thoroughly cooked before consumption.
- Reducing the risk of human-to-human transmission in the community arising from direct or close contact with infected patients, particularly with their body fluids. Close physical contact with Marburg patients should be avoided. Gloves and appropriate personal protective equipment should be worn when taking care of ill patients at home. Regular hand washing should be performed after visiting sick relatives in hospital, as well as after taking care of ill patients at home.
- Communities affected by Marburg should make efforts to ensure that the population is well informed, both about the nature of the disease itself and about necessary outbreak containment measures.
- Outbreak containment measures include prompt and safe burial of the dead, identifying people who may have been in contact with someone infected with Marburg and monitoring their health for 21 days, separating the healthy from the sick to prevent further spread, and maintaining good hygiene and a clean environment need to be observed.
- Reducing the risk of possible sexual transmission. Based on further analysis of ongoing research, WHO recommends that male survivors of Marburg virus disease practice safe sex and hygiene for 12 months from onset of symptoms or until their semen twice tests negative for Marburg virus. Contact with body fluids should be avoided and washing with soap and water is recommended. WHO does not recommend isolation of male or female convalescent patients whose blood has been tested negative for Marburg virus.
Controlling infection in healthcare settings
Healthcare workers should always take standard precautions when caring for patients, regardless of their presumed diagnosis. These include basic hand hygiene, respiratory hygiene, use of personal protective equipment (to block splashes or other contact with infected materials), safe injection practices and safe and dignified burial practices.
Healthcare workers caring for patients with suspected or confirmed Marburg virus should apply extra infection control measures to prevent contact with the patient’s blood and body fluids and contaminated surfaces or materials such as clothing and bedding. When in close contact (within 1 metre) of patients with MVD, health-care workers should wear face protection (a face shield or a medical mask and goggles), a clean, non-sterile long-sleeved gown, and gloves (sterile gloves for some procedures).
Laboratory workers are also at risk. Samples taken from humans and animals for investigation of Marburg infection should be handled by trained staff and processed in suitably equipped laboratories.
WHO response
WHO aims to prevent Marburg outbreaks by maintaining surveillance for Marburg virus disease and supporting at-risk countries to develop preparedness plans. The following document provides overall guidance for control of Ebola and Marburg virus outbreaks:
When an outbreak is detected WHO responds by supporting surveillance, community engagement, case management, laboratory services, contact tracing, infection control, logistical support and training and assistance with safe burial practices.
WHO has developed detailed advice on Marburg infection prevention and control:
Table: Chronology of major Marburg virus disease outbreaks
| Year |
Country |
Cases |
Deaths |
Case fatality Rate |
| 2014 |
Uganda |
1 |
1 |
100% |
| 2012 |
Uganda |
15 |
4 |
27% |
| 2008 |
Netherland (ex-Uganda) |
1 |
1 |
100% |
| 2008 |
United States of America (ex-Uganda) |
1 |
0 |
0% |
| 2007 |
Uganda |
4 |
2 |
50% |
| 2005 |
Angola |
374 |
329 |
88% |
| 1998 to 2000 |
Democratic Republic of the Congo |
154 |
128 |
83% |
| 1987 |
Kenya |
1 |
1 |
100% |
| 1980 |
Kenya |
2 |
1 |
50% |
| 1975 |
South Africa |
3 |
1 |
33% |
| 1967 |
Yugoslavia |
2 |
0 |
0% |
| 1967 |
Germany |
29 |
7 |
24% |