Outbreak of Seoul Virus Among Rats and Rat Owners — United States and Canada, 2017
February 4th, 2018Kerins JL, Koske SE, Kazmierczak J, et al. Outbreak of Seoul Virus Among Rats and Rat Owners — United States and Canada, 2017. MMWR Morb Mortal Wkly Rep 2018;67:131–134. DOI: http://dx.doi.org/10.15585/mmwr.mm6704a5.
In December 2016, the Wisconsin Department of Health Services (WDHS) notified CDC of a patient hospitalized with fever, leukopenia, elevated transaminases, and proteinuria. The patient owned and operated an in-home rattery, or rat-breeding facility, with approximately 100 Norway rats, primarily bred as pets. A family member developed similar symptoms 4 weeks later, but was not hospitalized. Because both patients were known to have rodent contact, they were tested for hantavirus infections. In January 2017, CDC confirmed recent, acute Seoul virus infection in both patients. An investigation was conducted to identify additional human and rat infections and prevent further transmission. Ultimately, the investigation identified 31 facilities in 11 states with human and/or rat Seoul virus infections; six facilities also reported exchanging rats with Canadian ratteries. Testing of serum samples from 183 persons in the United States and Canada identified 24 (13.1%) with Seoul virus antibodies; three (12.5%) were hospitalized and no deaths occurred. This investigation, including cases described in a previously published report from Tennessee (1), identified the first known transmission of Seoul virus from pet rats to humans in the United States and Canada. Pet rat owners should practice safe rodent handling to prevent Seoul virus infection (2).
Seoul virus is an Old World hantavirus in the Bunyaviridae family. Its natural reservoir is the Norway rat (Rattus norvegicus). Rats infected with Seoul virus are asymptomatic, but can transmit the virus to humans through infectious saliva, urine, droppings, or aerosolization from contaminated bedding. Human signs and symptoms range from mild influenza-like illness to hemorrhagic fever with renal syndrome (HFRS). HFRS causes acute renal failure and can result in death; however, asymptomatic Seoul virus infections also occur. Wild Norway rats in the United States have been known to harbor Seoul virus infection (3), but transmission to humans is rare (4). Seoul virus is not known to spread from person to person. In the United Kingdom, Seoul virus transmission has occurred from pet rats to humans (5), but before this outbreak, infections had not been reported in pet rats in the United States or Canada.
Investigation and Results
After confirming Seoul virus infection in the Wisconsin patients, CDC and WDHS initiated investigations into rat shipments to (trace-back) and from (trace-forward) the rattery to identify suspected and confirmed facilities. Trace-back investigations initially extended back 2 months prior to onset of clinical disease, based on the known maximum incubation period for Seoul virus in humans. As additional confirmed facilities were identified, tracing focused instead on interactions with known infected facilities, sometimes as much as 1 year prior. Suspected facilities included ratteries, homes, or pet stores that sold rats to a confirmed facility (a facility where at least one human or rat tested positive for Seoul virus infection) or housed rats that lived at or comingled with rats from a confirmed facility. Once a suspected facility was identified, local or state health officials interviewed persons with a history of rodent contact associated with the facility about their rat exposure and health history. Additionally, the primary rodent caretaker was interviewed using a standardized questionnaire to identify movement of rats into and out of the facility, including dates and locations where the rats were obtained. Local or state health officials offered laboratory testing for Seoul virus infection to all persons with rodent contact. Officials recommended testing for persons with a history of febrile illness and exposure to rats from a confirmed facility and for rats at suspected and confirmed facilities. Trace-forward and trace-back investigations of rat shipments at confirmed facilities identified additional suspected facilities, which were similarly assessed.
A suspected human case of Seoul virus infection was defined as a febrile illness (recorded temperature >101°F [38.3°C] or subjective history of fever) or an illness clinically compatible with Seoul virus infection (myalgia, headache, renal failure, conjunctival redness, thrombocytopenia, or proteinuria) without laboratory confirmation in a person reporting contact with rats from a confirmed or suspected facility. Human Seoul virus infections were laboratory-confirmed by detection of Seoul virus-specific immunoglobulin M (IgM) and/or immunoglobulin G (IgG) (6) antibodies by enzyme-linked immunosorbent assay (ELISA). In the United States, Seoul virus infections in rats were confirmed through detection of viral RNA by reverse transcription–polymerase chain reaction (RT-PCR) and/or IgG ELISA at CDC, or by CDC-validated commercial IgG testing. In Canada, public health officials investigated rat breeding facilities that exported rats to and imported rats from affected U.S. facilities. Seoul virus infection was detected in Canadian rats from breeding facilities using the same serologic and molecular-based protocols described for United States facilities.
By March 16, 2017, trace-forward and trace-back investigations identified approximately 100 suspected facilities in 21 states. Among these, 31 facilities in 11 states* had laboratory-confirmed human or rat infections, including a previously reported household in Tennessee with two confirmed human infections (1). Six confirmed facilities in six states (Georgia, Illinois, Missouri, South Carolina, Tennessee, and Utah) reported exchanging rats with Canadian ratteries during their trace-forward and trace-back investigations. A total of 163 persons in the United States and 20 in Canada consented to serologic testing; 17 (10.4%) U.S. residents and one (5.0%) Canadian resident had detectable IgM and IgG antibodies, indicating recent infection, and four (2.5%) U.S. residents and two (10.0%) Canadian residents had only IgG antibodies, indicating past or convalescent infection. Among the 17 U.S. patients with recent Seoul virus infection, eight reported recent febrile illness. Three were hospitalized, but did not develop HFRS, and all recovered. Serious illness was not reported in any Canadian patients. All strains detected in Canada and the United States were indistinguishable from one another based on nucleotide sequencing (7), indicating that a single strain was responsible for the outbreak. No single facility was identified as the origin of the outbreak.
Public Health Response
On January 24, CDC issued a Health Alert Notice to notify health departments and health care providers of the Seoul virus investigations.† On February 20, the World Health Organization was notified of the U.S. and Canadian infections and investigations as required by International Health Regulations.§ On January 31 and May 9, 2017, CDC and the Pet Industry Joint Advisory Council hosted calls to provide updates on the Seoul virus outbreak and to answer questions for the pet industry and fancy rat community. CDC created a website with Seoul virus facts and frequently asked questions for the public.
Health departments notified suspected and confirmed facilities and placed those facilities under quarantine, allowing no rats to enter or leave. Rat contact was limited to as few persons as possible to reduce transmission. In suspected facilities, CDC recommended rat testing be performed under the supervision of a public health official or licensed veterinarian. The quarantine was lifted when at least 4 weeks had elapsed since the newest animal was introduced, and all rats subsequently tested negative. Rats belonging to owners who refused to test their animals could remain quarantined for life or be euthanized. CDC recommended euthanasia of all rats in confirmed facilities as the most effective method to prevent transmission, although control recommendations differed by state and country according to local policies and response capacities. If euthanasia was not possible, then owners could either quarantine all rats for life or pursue quarantine with testing and culling. The testing and culling strategy entailed testing all rats and euthanizing only infected rats. Testing and euthanasia were repeated at 4 week intervals until all rats tested negative and the quarantine was lifted. In Canada, public health officials opted for education and a voluntary testing and culling approach to control Seoul virus transmission.
Discussion
This outbreak report, in parallel to the previously described investigation in Tennessee (1), describes the first known cases of Seoul virus infection in humans attributable to contact with pet rats in the United States and Canada. Human hantavirus infections are nationally notifiable in the United States and suspected cases should be reported to state or local health departments. Health care providers should consider Seoul virus infection in patients with febrile illness who report rat exposure; CDC recommends testing for any person with compatible illness and rodent contact. Testing is available at CDC¶ and through some state and commercial laboratories. In Canada, testing is available for symptomatic persons with rat exposure, rattery owners associated with this investigation, and their rats through public health laboratories; for individually owned pet rats and ratteries not associated with the investigation, testing is available through a commercial laboratory.
Pet rat owners should be aware of the potential for Seoul virus infection. To keep themselves and their pets healthy, all persons with rodent contact should avoid bites or scratches and practice good hand hygiene, especially children and persons with compromised immune systems (2). CDC recommends hand washing after caring for rodents and before eating, drinking, or preparing food (2). If a pet rat is suspected of having Seoul virus, the person cleaning the rodent environment should wear a respirator, gloves, and cover any scratches or open wounds (8). An adult should routinely disinfect rat cages and accessories, including used bedding, with a 10% bleach solution or a commercial disinfectant (8). More information about rodent contact and disease prevention is available from CDC (8,9).
Rattery owners are encouraged to quarantine any newly acquired rats for 4 weeks and to test these rats for Seoul virus antibodies before allowing them to comingle with other rats. Commercial laboratories can perform Seoul virus testing of rodent blood samples, and comparisons of results from shared samples have been concordant with CDC’s ELISA and RT-PCR assays. To prevent transmission to humans, CDC recommends euthanasia of all rats in facilities with human or rat Seoul virus infections. Further guidance on methods to eradicate Seoul virus from infected ratteries should be obtained from local or state health departments.
References
- Fill MA, Mullins H, May AS, et al. Notes from the field: multiple cases of Seoul virus infection in a household with infected pet rats—Tennessee, December 2016–April 2017. MMWR Morb Mortal Wkly Rep 2017;66:1081–2. CrossRef PubMed
- CDC. Key messages about pet rodents. Atlanta, GA: US Department of Health and Human Services, CDC; 2017. https://www.cdc.gov/healthypets/pets/small-mammals/petrodents.html
- Childs JE, Korch GW, Smith GA, Terry AD, Leduc JW. Geographical distribution and age related prevalence of antibody to Hantaan-like virus in rat populations of Baltimore, Maryland, USA. Am J Trop Med Hyg 1985;34:385–7. CrossRef PubMed
- Knust B, Rollin PE. Twenty-year summary of surveillance for human hantavirus infections, United States. Emerg Infect Dis 2013;19:1934–7 . CrossRef PubMed
- Jameson LJ, Taori SK, Atkinson B, et al. Pet rats as a source of hantavirus in England and Wales, 2013. Euro Surveill 2013;18:20415. PubMed
- Ksiazek TG, West CP, Rollin PE, Jahrling PB, Peters CJ. ELISA for the detection of antibodies to Ebola viruses. J Infect Dis 1999;179(Suppl 1):S192–8. CrossRef PubMed
- Woods C, Palekar R, Kim P, et al. Domestically acquired Seoul virus causing hemorrhagic fever with renal syndrome—Maryland, 2008. Clin Infect Dis 2009;49:e109–12. CrossRef PubMed
- CDC. Cleaning up after pet rodents to reduce the risk of Seoul virus infection. Atlanta, GA: US Department of Health and Human Services, CDC; 2017. https://www.cdc.gov/hantavirus/outbreaks/seoul-virus/cleaning-up-pet-rodents.html
- CDC. Healthy pets healthy people. Atlanta, GA: US Department of Health and Human Services, CDC; 2017. https://www.cdc.gov/healthypets/
2017-2018 Influenza Season Week 4 ending January 27, 2018
February 3rd, 2018Synopsis:
During week 4 (January 21-27, 2018), influenza activity increased in the United States.
- Viral Surveillance: The most frequently identified influenza virus subtype reported by public health laboratories during week 4 was influenza A(H3). The percentage of respiratory specimens testing positive for influenza in clinical laboratories remained elevated.
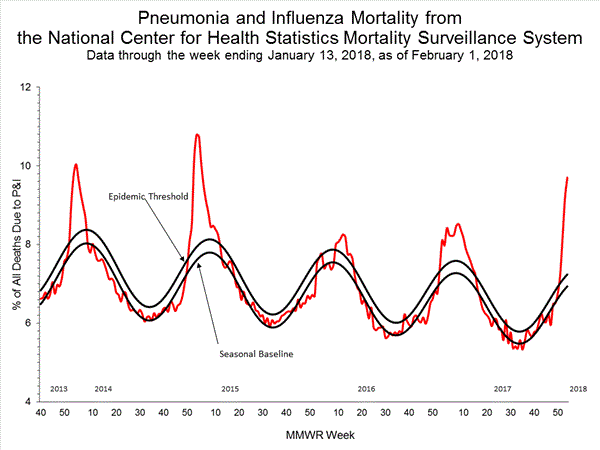
- Pneumonia and Influenza Mortality: The proportion of deaths attributed to pneumonia and influenza (P&I) was above the system-specific epidemic threshold in the National Center for Health Statistics (NCHS) Mortality Surveillance System.
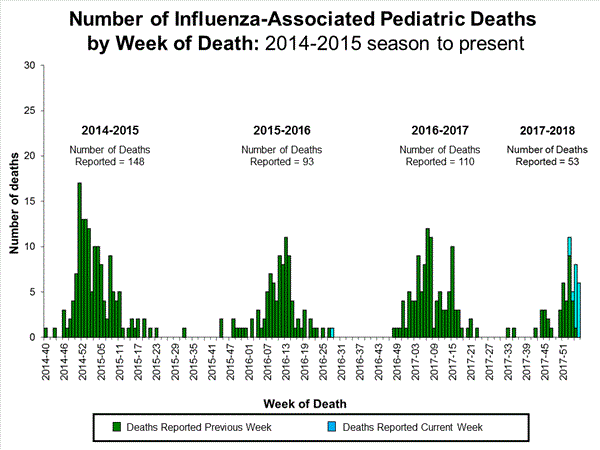
- Influenza-associated Pediatric Deaths: Seventeen influenza-associated pediatric deaths were reported, one of which occurred during the 2015-2016 season.
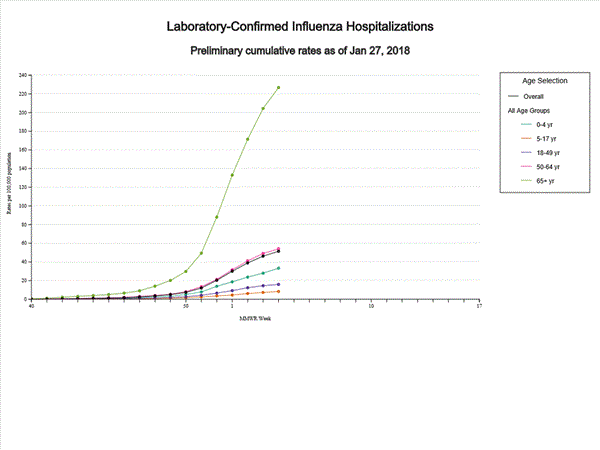
- Influenza-associated Hospitalizations: A cumulative rate of 51.4 laboratory-confirmed influenza-associated hospitalizations per 100,000 population was reported.
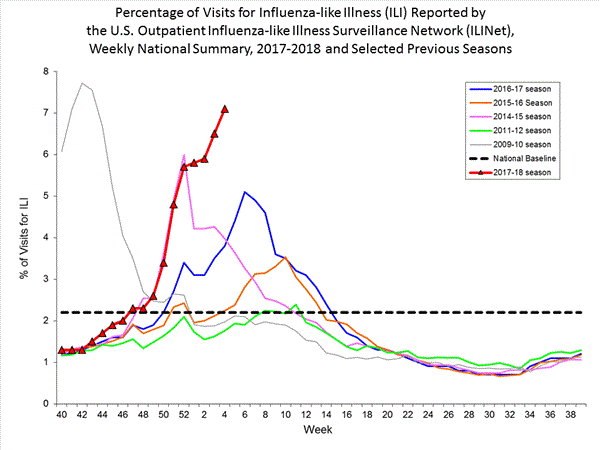
- Outpatient Illness Surveillance: The proportion of outpatient visits for influenza-like illness (ILI) was 7.1%, which is above the national baseline of 2.2%. All 10 regions reported ILI at or above region-specific baseline levels. New York City, the District of Columbia, and 42 states experienced high ILI activity; Puerto Rico and two states experienced moderate ILI activity; three states experienced low ILI activity; and three states experienced minimal ILI activity.
- Geographic Spread of Influenza:The geographic spread of influenza in Puerto Rico and 48 states was reported as widespread; Guam and one state reported regional activity; the District of Columbia and one state reported local activity; and the U.S. Virgin Islands reported sporadic activity.
Influenza-Associated Pediatric Mortality:
Seventeen influenza-associated pediatric deaths were reported to CDC during week 4.
Five deaths were associated with an influenza A(H3) virus and occurred during weeks 1, 2, 3, and 4 (the weeks ending January 6, January 13, January 20, and January 27, 2018). Two deaths were associated with an influenza A(H1N1)pdm09 virus and occurred during weeks 3 and 4 (the weeks ending January 20, 2018, and January 27, 2018, respectively). Four deaths were associated with an influenza A virus for which no subtyping was performed and occurred during weeks 3 and 4. Five deaths were associated with an influenza B virus and occurred during weeks 1, 3, and 4 (the week ending January 6, January 20, and January 27, 2018, respectively).
A total of 53 influenza-associated pediatric deaths have been reported for the 2017-2018 season.
One death that occurred during the 2015-2016 season was associated with an influenza A virus for which no subtyping was performed and occurred during week 28 (the week ending July 16, 2016). This death brings the total number of reported influenza-associated deaths occurring during that season to 93.
Additional data can be found at: http://gis.cdc.gov/GRASP/Fluview/PedFluDeath.html.
Influenza-Associated Hospitalizations:
The Influenza Hospitalization Surveillance Network (FluSurv-NET) conducts population-based surveillance for laboratory-confirmed influenza-related hospitalizations in children younger than 18 years of age (since the 2003-2004 influenza season) and adults (since the 2005-2006 influenza season).
The FluSurv-NET covers more than 70 counties in the 10 Emerging Infections Program (EIP) states (CA, CO, CT, GA, MD, MN, NM, NY, OR, and TN) and additional Influenza Hospitalization Surveillance Project (IHSP) states. The IHSP began during the 2009-2010 season to enhance surveillance during the 2009 H1N1 pandemic. IHSP sites included IA, ID, MI, OK and SD during the 2009-2010 season; ID, MI, OH, OK, RI, and UT during the 2010-2011 season; MI, OH, RI, and UT during the 2011-2012 season; IA, MI, OH, RI, and UT during the 2012-2013 season; and MI, OH, and UT during the 2013-2014, 2014-15, 2015-16, 2016-17, and 2017-18 seasons.
Data gathered are used to estimate age-specific hospitalization rates on a weekly basis, and describe characteristics of persons hospitalized with influenza illness. The rates provided are likely to be an underestimate as influenza-related hospitalizations can be missed, either because testing is not performed, or because cases may be attributed to other causes of pneumonia or other common influenza-related complications.
A total of 14,676 laboratory-confirmed influenza-associated hospitalizations were reported between October 1, 2017 and January 27, 2018. The overall hospitalization rate was 51.4 per 100,000 population. The highest rate of hospitalization was among adults aged ≥65 years (226.8 per 100,000 population), followed by adults aged 50-64 (54.0 per 100,000 population) and children aged 0-4 years (33.3 per 100,000 population). Among 14,676 hospitalizations, 12,849 (87.5%) were associated with influenza A virus, 1,762 (12.0%) with influenza B virus, 35 (0.2%) with influenza A virus and influenza B virus co-infection, and 30 (0.2%) with influenza virus for which the type was not determined. Among those with influenza A subtype information, 2,797 (86.5%) were A(H3N2) and 437 (13.5%) were A(H1N1)pdm09 virus.
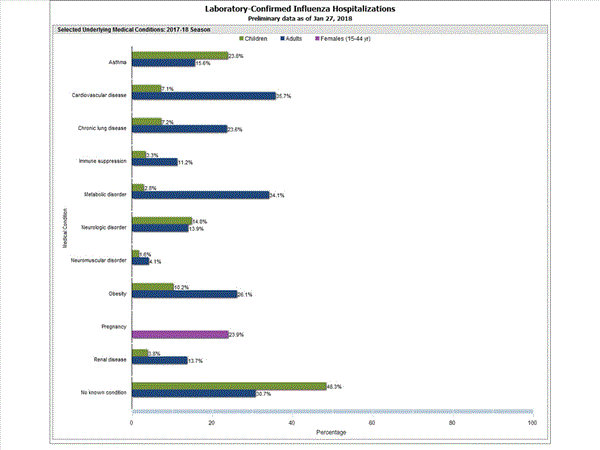
Among 1,708 hospitalized adults with information on underlying medical conditions, 1,183 (69.3%) had at least one reported underlying medical condition; the most commonly reported were cardiovascular disease, metabolic disorder, obesity, and chronic lung disease. Among 180 hospitalized children with information on underlying medical conditions, 93 (51.7%) had at least one underlying medical condition; the most commonly reported were asthma, neurologic disorder, and obesity. Among 138 hospitalized women of childbearing age (15-44 years) with information on pregnancy status, 33 (23.9%) were pregnant.
Additional FluSurv-NET data can be found at: http://gis.cdc.gov/GRASP/Fluview/FluHospRates.html and http://gis.cdc.gov/grasp/fluview/FluHospChars.html.
Outpatient Illness Surveillance:
Nationwide during week 4, 7.1% of patient visits reported through the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet) were due to influenza-like illness (ILI). This percentage is above the national baseline of 2.2%.(ILI is defined as fever (temperature of 100°F [37.8°C] or greater) and cough and/or sore throat.)
Additional ILINet data, including national, regional and select state-level data, are available at http://gis.cdc.gov/grasp/fluview/fluportaldashboard.html.
On a regional level, the percentage of outpatient visits for ILI ranged from 2.8% to 13.0% during week 4. All 10 regions reported percentages of outpatient visits for ILI at or above their region specific baselines.
From 1 Jul 2017-30 Jan 2018, 213 yellow fever cases were confirmed in Brazil, including 81 deaths.
February 2nd, 2018A midseason glimpse of flu vaccine effectiveness (VE) in Canada shows that protection against the H3N2 strain is very low, similar to what Australian scientists reported in the fall
February 2nd, 2018“….As reported from Australia for the 2017 southern hemisphere vaccine, interim estimates from Canada for the 2017/18 northern hemisphere vaccine indicate low VE of less than 20% against influenza A(H3N2), notably among working-age adults. While the influenza A(H3N2) epidemic continues, adjunct protective measures should be reinforced to minimise the associated disease burden in high-risk individuals….”
CDC is dramatically downsizing its epidemic prevention activities in 39 out of 49 countries because money is running out
February 2nd, 2018“…..The CDC plans to narrow its focus to 10 “priority countries,” starting in October 2019, the official said. They are India, Thailand and Vietnam in Asia; Jordan in the Middle East; Kenya, Uganda, Liberia, Nigeria and Senegal in Africa; and Guatemala in Central America.
Countries where the CDC is planning to scale back include some of the world’s hot spots for emerging infectious disease, such as China, Pakistan, Haiti, Rwanda and Congo……”
India: Wanting to Give Half a Billion People Free Health Care
February 1st, 2018“…..The health care plan, part of the government’s 2018-19 budget presented on Thursday, would offer 100 million families up to 500,000 rupees, or about $7,860, of coverage each year. That sum, while small by Western standards, would be enough to cover the equivalent of five heart surgeries in India…..”
1918 H1N1 & cytokine responses in ferrets: Could explain the severity of disease….
February 1st, 20181918 H1N1 influenza virus replicates and induces pro-inflammatory cytokine responses in extra-respiratory tissues of ferrets.
The Journal of Infectious Diseases, jiy003, https://doi.org/10.1093/infdis/jiy003
Published: 10 January 2018
“….1918 H1N1 virus spread to, and induced cytokine responses in tissues outside the respiratory tract, which likely contributed to the severity of infection. Moreover, our data support the suggested link between 1918 H1N1 infection and CNS disease.….”